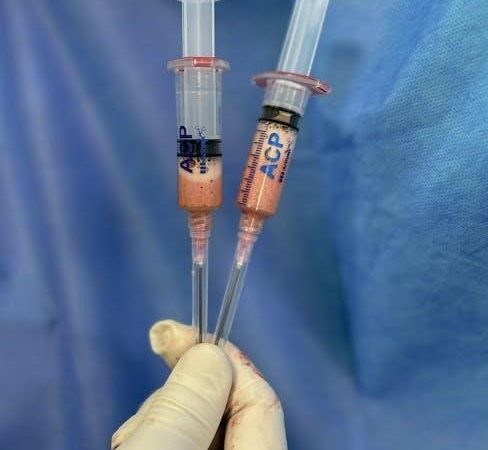
Platelet-Rich Plasma (PRP) harnesses the body’s natural healing mechanisms, utilizing concentrated platelets from a small blood sample. These vital cells initiate repair and regeneration.
What is Platelet-Rich Plasma?
Platelet-Rich Plasma (PRP) is an autologous concentration of platelets in a small volume of plasma, prepared from whole blood. It’s not simply whole blood; it contains a significantly higher concentration of platelets – typically three to nine times greater – than what is normally found in circulation.
These platelets, crucial for blood clotting and wound healing, release growth factors and cytokines that stimulate cellular repair. Platelets, or thrombocytes, are essential for hemostasis, responding to vascular injury by forming clots. They are tiny cellular fragments produced in the bone marrow, acting as first responders to breaches in vessel walls.
The Role of Platelets in Healing
Platelets are fundamental to the body’s natural healing cascade, extending far beyond simple blood clotting. Upon activation, these cellular fragments release a plethora of growth factors – proteins stimulating cell growth, proliferation, and differentiation. Proteins on platelet walls aid adhesion to blood vessels, extending filaments during clotting.
This initiates tissue repair, collagen synthesis, and angiogenesis (new blood vessel formation). A normal platelet count (150,000-450,000/µL) ensures adequate response to injury, while low counts can impair healing and increase bleeding risk.

PRP Preparation Protocol: A Comprehensive Overview
PRP preparation involves drawing a patient’s blood, processing it to concentrate platelets, and then activating the enriched plasma for therapeutic applications.
Patient Selection Criteria
Ideal candidates for PRP therapy generally exhibit good overall health, though specific criteria depend on the intended application. Patients with active infections, severe systemic diseases, or bleeding disorders may be excluded. A thorough medical history and physical examination are crucial.
Evaluating platelet counts is essential; levels outside the normal range (150,000-450,000/µL) can impact PRP effectiveness. Medications affecting platelet function, like anticoagulants, require careful consideration and potential temporary discontinuation. Realistic patient expectations regarding treatment outcomes are also vital for satisfaction.
Anticoagulation Considerations
Anticoagulant medications significantly impact PRP preparation, potentially compromising platelet concentration and function. Heparin, warfarin, and direct oral anticoagulants (DOACs) interfere with clot formation, necessitating careful management. Temporary cessation of these medications, guided by a physician, is often required before PRP collection.
The duration of discontinuation varies based on the specific anticoagulant and patient risk factors. Alternative anticoagulation strategies or bridging therapies might be considered. Thorough documentation of medication history is paramount to ensure optimal PRP quality and therapeutic efficacy.

Centrifugation Process: The Core of PRP Preparation
Centrifugation separates blood components, concentrating platelets into PRP. This crucial step utilizes centrifugal force to isolate platelets from red and white blood cells.
Centrifuge Types and Settings
Centrifugation relies on specialized equipment, with both benchtop and refrigerated centrifuges commonly employed for PRP preparation. Benchtop models offer convenience, while refrigerated units maintain optimal platelet viability. Settings are critical; two-stage centrifugation is frequently utilized.
The initial, slower spin separates red blood cells, followed by a higher-speed spin to concentrate platelets. Precise speed (typically 1500-3000 RPM) and duration (ranging from 10-30 minutes) are vital for optimal PRP yield and quality, varying based on the chosen system and desired platelet concentration.
Centrifugation Speed and Duration
Optimal centrifugation parameters are crucial for effective PRP preparation. Generally, a two-step process is preferred, beginning with a lower speed (around 1500-2000 RPM) for 10-15 minutes to separate red blood cells. This prevents contamination and ensures a clearer PRP layer.
Subsequently, a higher speed (2500-3500 RPM) for another 10-20 minutes concentrates the platelets. Variations exist based on centrifuge type and desired platelet concentration; precise timing and speed are essential for consistent results.
Layer Separation and Component Identification
Following centrifugation, distinct layers form within the tube. The bottom layer consists of concentrated red blood cells, while the middle layer contains the platelet-rich plasma – a clear, yellowish fluid. Above this lies the platelet-poor plasma (PPP), appearing paler in color.
A buffy coat, containing white blood cells, may also be visible. Careful aspiration of the PRP layer, avoiding contamination from RBCs or PPP, is vital for a high-quality PRP product. Accurate identification of each layer is paramount.
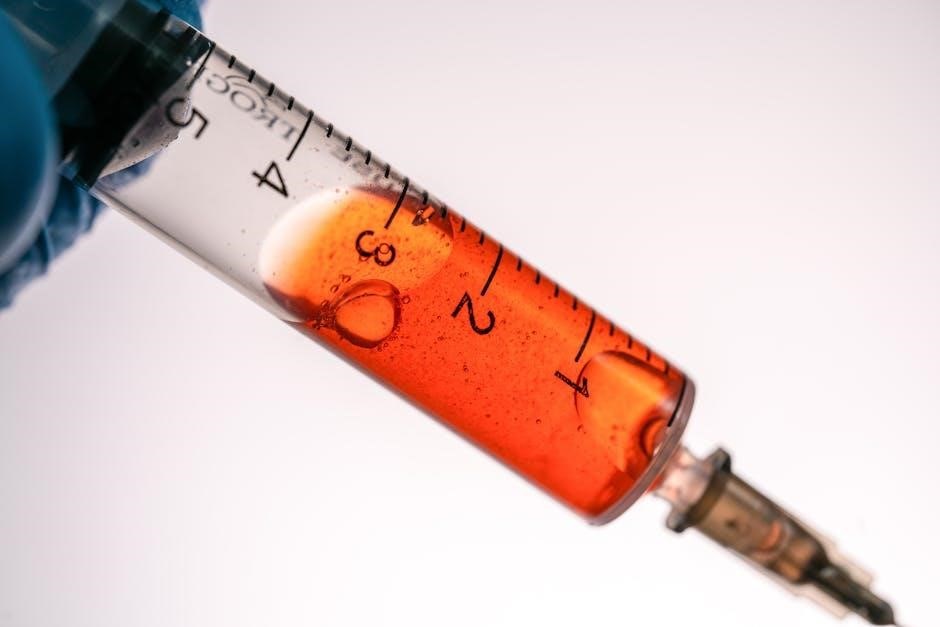
PRP Component Analysis
Rigorous analysis confirms PRP quality. Platelet counts, WBC concentration, and RBC contamination are assessed to ensure therapeutic efficacy and patient safety standards are met.
Platelet Count Verification
Accurate platelet quantification is paramount. A normal platelet range is typically 150,000 to 450,000 per microliter of blood; however, PRP aims for a significantly higher concentration. Automated hematology analyzers, or manual hemocytometer counting, are employed to verify the platelet count within the prepared PRP sample. This ensures the final product contains sufficient platelets—often 3-7 times baseline—to stimulate the desired biological response. Consistent and reliable platelet counts are crucial for predictable clinical outcomes and standardized PRP protocols.
White Blood Cell (WBC) Concentration
WBCs within PRP contribute to the inflammatory phase of healing, releasing growth factors and cytokines. However, excessive WBCs can induce unwanted inflammation. Therefore, monitoring WBC concentration is vital. PRP preparations can be leukocyte-rich or leukocyte-poor, depending on the clinical application. Automated cell counters determine WBC levels, typically expressed as cells per microliter. Balancing the beneficial inflammatory response with minimizing potential adverse effects requires careful control of WBC concentration during PRP preparation.
Red Blood Cell (RBC) Contamination Assessment
RBC contamination in PRP is generally undesirable, potentially triggering an inflammatory response and hindering the healing process. Minimal RBC presence is crucial for optimal PRP efficacy. Visual inspection post-centrifugation can reveal noticeable RBC contamination, appearing as a reddish hue. Automated hematology analyzers provide precise RBC counts. Maintaining a low RBC concentration—typically less than 5%—is a key quality control parameter. Careful layering during collection and centrifugation minimizes RBC carryover into the final PRP product.
Activation Methods for PRP
PRP activation is essential to initiate the platelet cascade, releasing growth factors for tissue repair. Common methods include calcium chloride, thrombin, or UV irradiation.
Calcium Chloride Activation
Calcium chloride is a frequently employed activator for PRP, triggering rapid platelet degranulation and subsequent release of growth factors. This method involves adding a small amount of calcium chloride directly to the prepared PRP, initiating the coagulation cascade and platelet activation. It’s a relatively simple and cost-effective technique, widely used in various clinical applications. However, careful dosage is crucial; excessive calcium chloride can lead to premature and uncontrolled clotting. The concentration typically ranges from 10% to 20%, ensuring optimal activation without compromising the PRP’s therapeutic potential. This method provides a reliable and predictable activation pathway for enhanced healing responses.
Thrombin Activation
Thrombin represents a potent activator for PRP, mimicking the body’s natural coagulation process with exceptional efficiency. Introducing thrombin to PRP initiates a swift and robust platelet activation, leading to a substantial release of growth factors crucial for tissue repair. While highly effective, thrombin activation demands precise control due to its potent nature. Overuse can result in rapid clot formation, potentially diminishing the PRP’s bioavailability. Typically, low concentrations are utilized to ensure controlled activation and sustained growth factor release, optimizing therapeutic outcomes in diverse clinical scenarios.
UV Irradiation Activation
UV irradiation presents an alternative method for PRP activation, utilizing ultraviolet light to stimulate platelet degranulation and growth factor release. This technique offers a potentially sterile activation pathway, minimizing the risk of contamination compared to other methods. However, careful calibration of UV exposure is paramount; excessive irradiation can damage platelet components and diminish their regenerative capacity. The process requires specialized equipment and precise protocols to ensure optimal activation without compromising PRP quality, making it a less commonly used, yet viable, activation strategy.
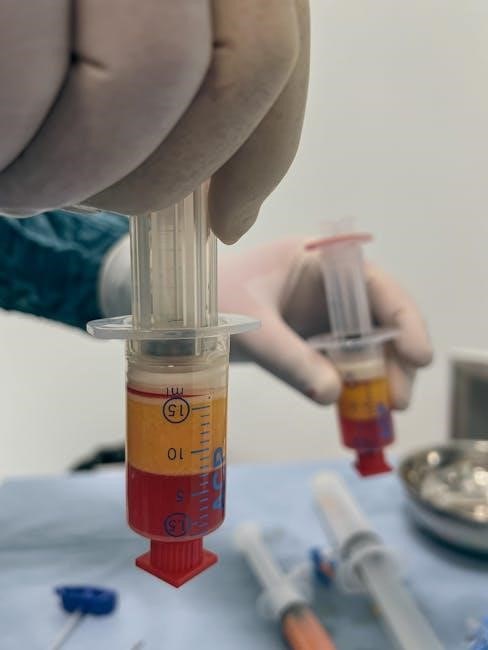
Quality Control and Storage
Rigorous quality control, including sterility checks and platelet counts, is essential. Proper storage—typically refrigerated—maintains PRP viability for a limited duration.
Sterility Assurance
Maintaining absolute sterility throughout the PRP preparation process is paramount to patient safety. All equipment, including centrifuge tubes, syringes, and collection containers, must be sterile and single-use. Aseptic technique, adhering to strict protocols, should be followed diligently by trained personnel during blood draw, centrifugation, and component handling. Regular monitoring of the preparation environment and confirmation of sterilization procedures are crucial. Any breach in sterility compromises the PRP’s therapeutic potential and introduces significant infection risks, potentially leading to adverse patient outcomes.
Storage Temperature and Duration
Proper storage is critical for maintaining PRP viability and effectiveness. Ideally, prepared PRP should be used immediately after processing to maximize platelet functionality. If immediate use isn’t feasible, refrigeration at 1-4°C (33.8-39.2°F) is recommended. However, prolonged storage diminishes platelet activation capacity. Generally, PRP is considered stable for up to 5-7 days under refrigerated conditions, though optimal results are achieved with same-day application. Freezing is generally discouraged as it can irreversibly damage platelets.

Clinical Applications of Prepared PRP
PRP’s regenerative properties find diverse applications, notably in orthopedics for joint and tendon injuries, dermatology for skin rejuvenation, and wound care acceleration.
Orthopedic Applications
PRP demonstrates significant promise in treating various orthopedic conditions. It’s frequently utilized for managing osteoarthritis, aiming to reduce pain and improve joint function by promoting cartilage repair. Tendon injuries, like tennis elbow or Achilles tendinopathy, also benefit from PRP’s regenerative capabilities, accelerating healing and strengthening tissues. Ligament sprains and muscle strains represent further applications, leveraging the growth factors within PRP to enhance recovery. Furthermore, PRP is explored post-surgically to aid in tissue healing and reduce inflammation, potentially improving long-term outcomes for patients undergoing orthopedic procedures.
Dermatological Applications
PRP is increasingly popular in dermatology for its rejuvenating effects on skin. It’s used to address acne scarring, stimulating collagen production to improve skin texture and reduce the appearance of blemishes. PRP also shows efficacy in treating age-related skin changes, like wrinkles and fine lines, promoting a more youthful complexion. Hair restoration is another key application, as PRP can stimulate hair follicles, potentially increasing hair density. Furthermore, PRP aids in wound healing, accelerating tissue repair and minimizing scarring post-procedures or injuries.
Wound Healing Applications
PRP significantly accelerates wound closure and improves tissue regeneration due to its concentrated growth factors. Chronic wounds, like diabetic ulcers and pressure sores, benefit from PRP’s ability to stimulate cellular proliferation and angiogenesis – the formation of new blood vessels. Surgical wounds also experience faster healing times and reduced scarring with PRP application. The growth factors within PRP recruit cells to the wound site, promoting collagen synthesis and extracellular matrix deposition, essential for robust tissue repair and minimizing complications.
Potential Complications and Risks
While generally safe, PRP carries minor risks like infection at the injection site, potential for thrombosis, and rare allergic reactions to components.
Infection Risk
Although uncommon, infection represents a potential complication following PRP procedures. Strict adherence to sterile techniques during preparation and injection is paramount to minimize this risk. Thorough skin disinfection before injection, alongside the use of sterile equipment and a sterile field, are crucial preventative measures.
Post-procedure, patients should monitor for signs of infection, including increased pain, redness, swelling, warmth, or pus at the injection site. Prompt medical attention is necessary if any of these symptoms develop, potentially requiring antibiotic treatment to effectively manage the infection and ensure optimal healing.
Thrombosis Risk
PRP, by its nature, concentrates platelets, inherently raising a theoretical risk of thrombosis – the formation of a blood clot. While generally low, this risk is heightened in individuals with pre-existing conditions like deep vein thrombosis (DVT) history or certain clotting disorders.
Careful patient evaluation is essential to identify those at increased risk. Avoiding PRP in patients with active thrombotic conditions is crucial. Post-procedure monitoring for signs of DVT, such as leg pain or swelling, is recommended, alongside encouraging adequate hydration and mobility.
Allergic Reaction Risk
Allergic reactions to PRP are considered rare, as the product is autologous – derived from the patient’s own blood. However, the risk isn’t entirely absent, particularly if any additives like calcium chloride or thrombin are used during the activation process.
Patients with known allergies to bovine-derived products (thrombin) should be carefully considered. Monitoring for signs of allergic reaction, including rash, itching, or difficulty breathing, is vital post-injection. Immediate access to emergency medical care is necessary should a reaction occur.
Future Directions in PRP Research
Ongoing research focuses on refining centrifugation, exploring novel activation methods, and investigating synergistic effects when PRP is combined with other regenerative therapies.
Optimizing Centrifugation Protocols
Current research endeavors aim to establish standardized centrifugation protocols for PRP preparation, acknowledging variability impacts final product quality. Investigating differing speeds, durations, and acceleration/deceleration rates is crucial. The goal is to maximize platelet concentration while minimizing red blood cell contamination and ensuring optimal platelet activation potential. Further studies will explore “soft spin” versus “hard spin” techniques, alongside closed versus open systems, to determine the most efficient and reproducible methods for consistently producing high-quality PRP for diverse clinical applications.
Investigating Novel Activation Techniques
Beyond traditional methods like calcium chloride and thrombin, researchers are exploring innovative PRP activation techniques. These include low-level laser therapy (LLLT) and focused ultrasound to stimulate platelet degranulation and growth factor release. Investigating the efficacy of these novel approaches is vital, aiming for enhanced and more targeted tissue regeneration. Studies focus on optimizing energy parameters and delivery methods to maximize biological response while minimizing potential cellular damage, ultimately improving clinical outcomes.

Exploring Combination Therapies
Synergistic effects are being investigated by combining PRP with other regenerative medicine approaches. This includes incorporating PRP with stem cells, growth factors, or biomaterials like hyaluronic acid or collagen scaffolds. These combinations aim to amplify therapeutic benefits, promoting faster and more complete tissue repair. Research focuses on identifying optimal ratios and delivery methods to maximize the combined regenerative potential, leading to improved outcomes in orthopedic, dermatological, and wound healing applications.